
27 Jul Is Retinol Banned in Europe?
Retinol is not getting banned in Europe, but new restrictions in the European Union will limit the use of retinol in skincare.
Is Europe banning the use of retinol in skincare? It seems that since the European Union notified the World Trade Organisation of its plan to regulate retinol use, it seems that retinol fans all around have been very concerned about the new restrictions and regulations. Here’s all you need to know about the new laws that will kick in to Europe.
Related blogpost:
New Zealand Bans “Forever Chemicals”/ PFAS in Cosmetics 2024
(I can hear fellow retinol fans around the world heave a sigh of relief.)
Retinol is one active ingredient that is evidenced based for improving acne and reducing signs of ageing like fine wrinkles and dark spots. You can learn more about retinols in How Does Retinol Work? Retinol Explained Simply.
IS RETINOL BANNED IN EUROPE?
No, retinol is NOT banned in the European Union. However, the European Union will limit retinol to 0.3% in face/hand products and to 0.05% in body products. This restriction will be implemented in end 2023; and the European Union has, as of June 8, 2023, notified the World Trade Organisation of a draft regulation of the change in the regulatory status of retinol and several other ingredients.
This restriction will also involve other retinoids that are also commonly found in skincare products such as retinyl palmitate, and retinyl acetate up to the concentrations of 0.3% and 0.05% of retinol equivalent in hand/face and body products respectively.
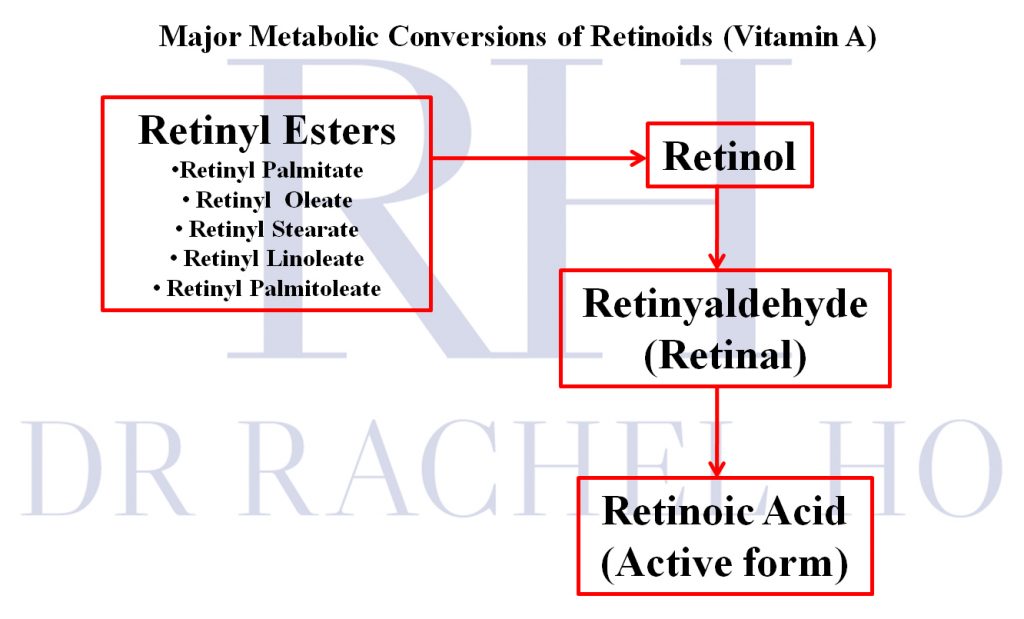
As explained in this blogpost Retinoids for Beginners, retinyl palmitate, and retinyl acetate are derivatives that undergo conversion to retinol and finally, retinoic acid in the skin. Retinoids are also known as vitamin A compounds. Vitamin A is also a micronutrient that can be obtained from our diet. Vitamin A is necessary for normal vision, the immune system, reproduction, and growth and development
How the family of retinoids are related.
REASONS FOR RETINOL BAN IN EUROPE
I’m certain that for so many retinol fans out there, this new restriction is perplexing. Why would the European Union ban, or more correctly, restrict the use of this popularly used anti-aging and anti-acne active ingredient?
Related blogpost:
What are Active Ingredients in Skincare & How do They Work?
This retinol restriction in the European Union was preceded by discussions within the European Union’s Scientific Committee on Consumer Safety (SCCS). Since 2012, the EU’s SCCS has raised concerns about the safety of retinoids. Between 2012 and 2013, the EU SCCS has worked with German and Norwegian authorities for safety assessments on vitamin A. in 2016 and in 2022, the EU SCCS published its opinions and recommendations to limit the concentration of retinols in cosmeceuticals to 0.3% in face/hand products; and to 0.05 in body lotions.
In its most recent recommendation that led to the EU adopting this retinol restriction, the SCCS concluded that aggregate exposure of vitamin A could exceed the upper limit determined by the European Food Safety Authority (EFSA). In other words, the total exposure to vitamin A from cosmetics, diet and nutritional supplements could exceed these safe limits; to cause fetal abnormalities (in pregnant women) and affect the liver and skin.
Interestingly, this new regulation does not involve retinaldehyde, which is metabolically closer to retinoic acid than retinol, retinyl palmitate, and retinyl acetate.
Vitamin A intake through diet and nutritional supplements exceeds intake via skincare.
Of relevance in this opinion is the proportion of retinoids in skin care that contributes to the total vitamin A intake because of absorption through the skin. In its statement, the EU’s SCCS recognises that “compared to food, the contribution of vitamin A from cosmetics is lower”. Nonetheless, the EU SCCS is concerned that this contribution of vitamin through skincare would still add to overall exposure for consumers at risk of the highest exposure from diet and food supplements.
Logically, for the EU to protect against unsafe exposures to vitamin A, it would make most sense for the EU to restrict the consumption of vitamin A from more significant sources i.e. diet and nutritional supplements. However, the EU SCCS provides opinions on health and safety risks of non-food consumer products (e.g. cosmetic products). Advising on the role of food and nutritional supplements is not within the purview of the EU SCCS.
More pregnancy do’s and don’t’s for skincare in this post.
DOES THIS MEAN THAT USING RETINOL IS DANGEROUS?
Not at all. In spite of this restriction by the European Union, the SCCS states its opinion that “vitamin A in cosmetics at the concentrations of 0.05% Retinol Equivalent (RE) in body lotion, and 0.3% RE for other leave-on and rinse-off products is safe”.
The purpose of this ban is to protect the most exposed consumers (which the SCCS considers to be 5% of the total population) who may already exceed the upper limit of vitamin A intake as determined by EFSA. The SCCS also recognises that “compared to food, the contribution of vitamin A from cosmetics is lower” and concluded that retinol products as such are safe.
While retinol use is not dangerous, retinol is not recommended for pregnancy. Retinol use is associated with retinoid dermatitis and purging. You can learn more about about retinol safety in these blogposts:
Pregnancy Skincare Guide: What’s Safe and What to Avoid
The Beginner’s Guide to Starting Retinoids
Is Your Skin Purging or Breaking Out?
WHAT ARE THE IMPLICATIONS OF THE RETINOL BAN/ RESTRICTION IN EUROPE
This draft by the EU has far reaching implications on the cosmeceuticals industry in the European Union as well public opinion of retinoids.
• Timelines
The European Commission plans to publish the regulation in the fourth quarter of 2023 officially; with adoption of this final ruling in June 2024. This draft is expected to set deadlines for implementing these new restrictions; and to allow the skincare industry lead time to adapt and avoid costs.
Based on current information, products that contain retinol, retinyl palmitate, and retinyl acetate, will have to meet these restrictions within 18-36 months. This means that by 2027, retinol products in the European Union and affected countries must abide by these new regulations- so no need to stock up on your favourite retinol products right now!
• Countries affected
Countries that will be affected by the European Union’s retinol restriction laws will include 27 countries of the European Union and countries that belong to the European Economic Area European Free Trade Association (Iceland, Liechtenstein, Norway, and North Macedonia). The United Kingdom and Switzerland are not members of the European Union; and are not bound by these new regulations.
Deadlines for new restrictions involving products with these ingredients by the EU Cosmetics Regulation. Image credit: Coslaw.
• Other affected ingredients/ products
The draft regulation by the European Union also includes other active ingredients. Although the restriction on retinol and retinoid products have received most attention in the media, the regulation implemented by the European Union actually also involves other active ingredients such as:
• Enzacamene or 4-Methylbenzylidene Camphor (a type of sunscreen filter)
• Kojic Acid
• Alpha-Arbutin and arbutin
• Triclocarban and
• Triclosan
WHICH POPULAR RETINOL PRODUCTS ARE/WILL BE AFFECTED BY THE RETINOL BAN/ RESTRICTION IN EUROPE?
Some popular and cult favourite retinol and related products will definitely be affected by this retinol ban/restriction in the European Union. As shared in my blogpost reviews of favourite and dud retinol products in my review of Popular Retinol Serums & Creams 2023 and in 2020; some well loved retinol serums and creams will have to be taken off the European markets.
These are the popular retinol serums and creams that contain more than 0.3% retinol; and will be affected by this retinol ban/ restriction:
• Paula’s Choice 1% Retinol Treatment
• The Ordinary Retinol 1% in Squalane
• SkinCeuticals Retinol 0.5
• Drunk Elephant A-Passioni™ Retinol Cream
I’ve reviewed the first three retinol serums in my blogposts Popular Retinol Serums & Creams 2023 and in 2020. I also share other retinol serums/ creams that you can use instead.
WHICH RETINOLS WILL REMAIN?
Some popular retinols that remain unaffected by this new regulation are:
• VICHY Liftactiv Retinol Serum (contains 0.2% retinol)
• CeraVe Resurfacing Retinol Serum (contains 0.3% retinol)
• Skinceuticals Retinol 0.3
• L’Oreal Paris Revitalift 0.3% Pure Retinol Night Serum
My reviews of popular retinol serums and creams are also on the blog.
WHAT ARE THE ALTERNATIVES TO RETINOL?
The retinol ban/ restriction in the European Union does not affect the other retinoids such as retinaldehyde, which is metabolically closer to retinoic acid than retinol,retinyl palmitate, and retinyl acetate. One consideration is to switch to retinaldehyde if your skin tolerates higher concentrations of retinol. One such product that I recommend is Avene A-OXitive NIGHT Peeling Cream, which contains 0.1% retinaldehyde; and you can read my review of it in this blogpost 5 Popular Retinol Serums & Creams Reviewed.
If you have acne; and aging related concerns such as wrinkles and dark spots, you can also consider retinoic acid (treinoin) and other prescription strength retinoids such as adaplene or trifrarotene (Aklief). Speak to your doctor to see if these options would be appropriate for you.
Related blogposts:
Acne: Types, Causes, Treatments and Tips for Prevention
5 Skincare Ingredients for Acne & Acne Prone Skin
Aklief (Trifarotene): 4th Generation Retinoid for Acne
You may also have come across skincare ingredients that claim to be “retinol alternatives”- examples are bakuchiol, rose hip oil and peptides. In Retinol Alternatives: Are They As Good as Retinol? I do a deep dive on the workings and evidence of these three “retinol alternatives”; and essentially although bakuchiol and rose hip may have some retinol like effects; each of them are also different from retinol.
CONCLUSION ON RETINOL BAN/ RESTRICTIONS IN THE EUROPEAN UNION
In conclusion, retinol is not being banned in the European Union. Rather, retinol use is being restricted to 0.3% in face and hand products and to 0.05% in body products. This is not because the EU deems that retinol use is dangerous. Instead, this restriction on retinol is out of an abundance of caution for users who already consume high amounts of vitamin A through diet and nutritional supplements; and additional use of retinols through skincare may cause them to exceed safety limits.
This restriction on retinol products is primed to start in 2024; but with a transition phase of up to 36 months. This means for readers residing in the affected European countries, retinol products exceeding 0.3% in the face/hands and 0.05% in the body will be discontinued by 2027. If you want to use retinol at higher concentrations, consider using retinaldehyde or other prescription strength retinoids, all of which are more potent than retinol.










No Comments